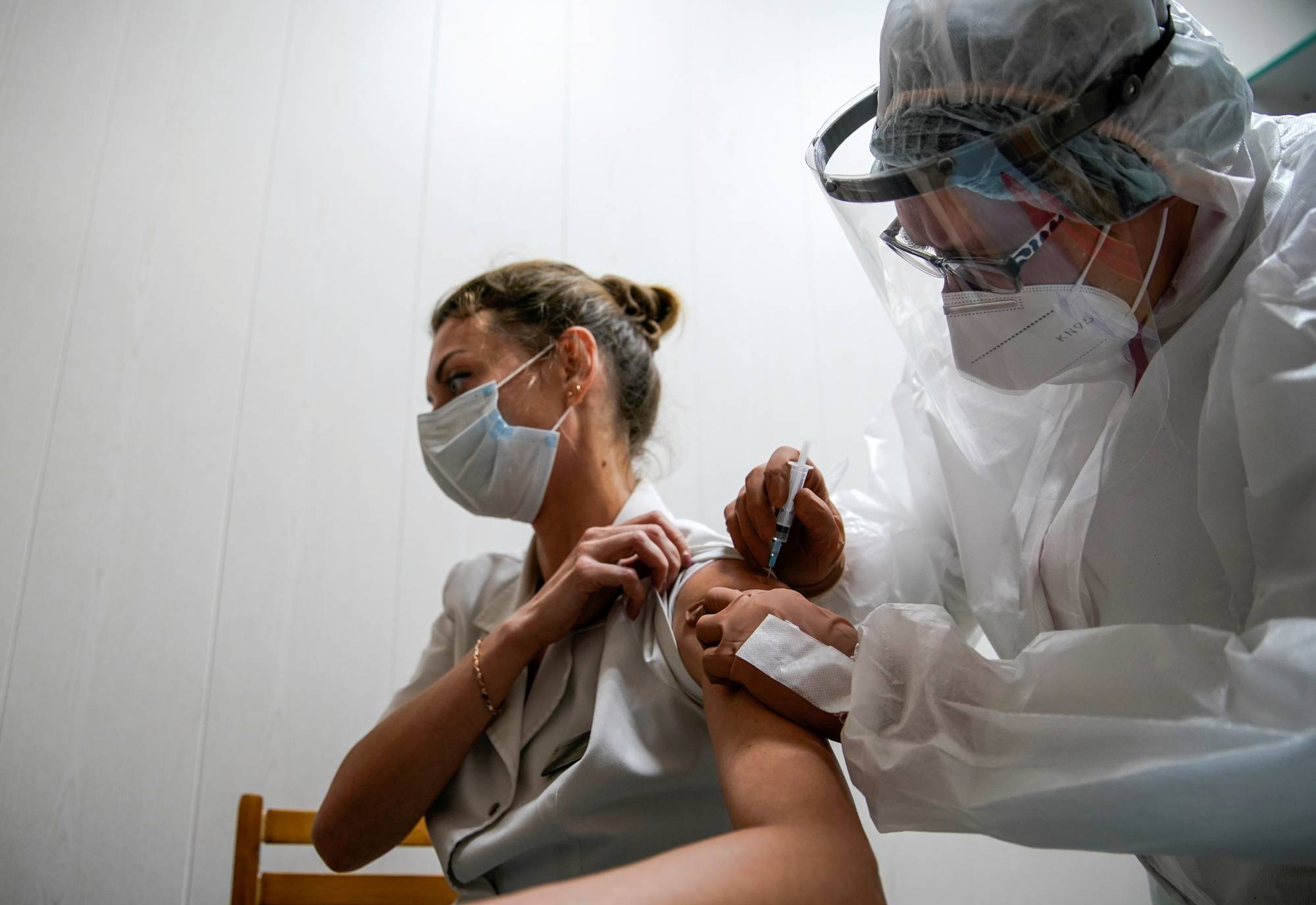
Eddie Rice is a believer in vaccines. The Melbourne locksmith has received jabs in the past and understands that they go through rigorous testing before they’re rolled out. This time, as researchers sprint ahead with potential shots to protect the world against COVID-19, he’s not so sure.
"This is a pretty unique one, just because it’s going to be so quick,” said Rice, 29. "I don’t know enough of the science to know 100% that it’s safe.”
Governments and drugmakers have long faced skepticism, and even hostility, from a small but vocal group of anti-vaccination campaigners. In the battle against the coronavirus, they may also run into reluctance from a broader swath of the population — people like Rice who would normally be on board.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.