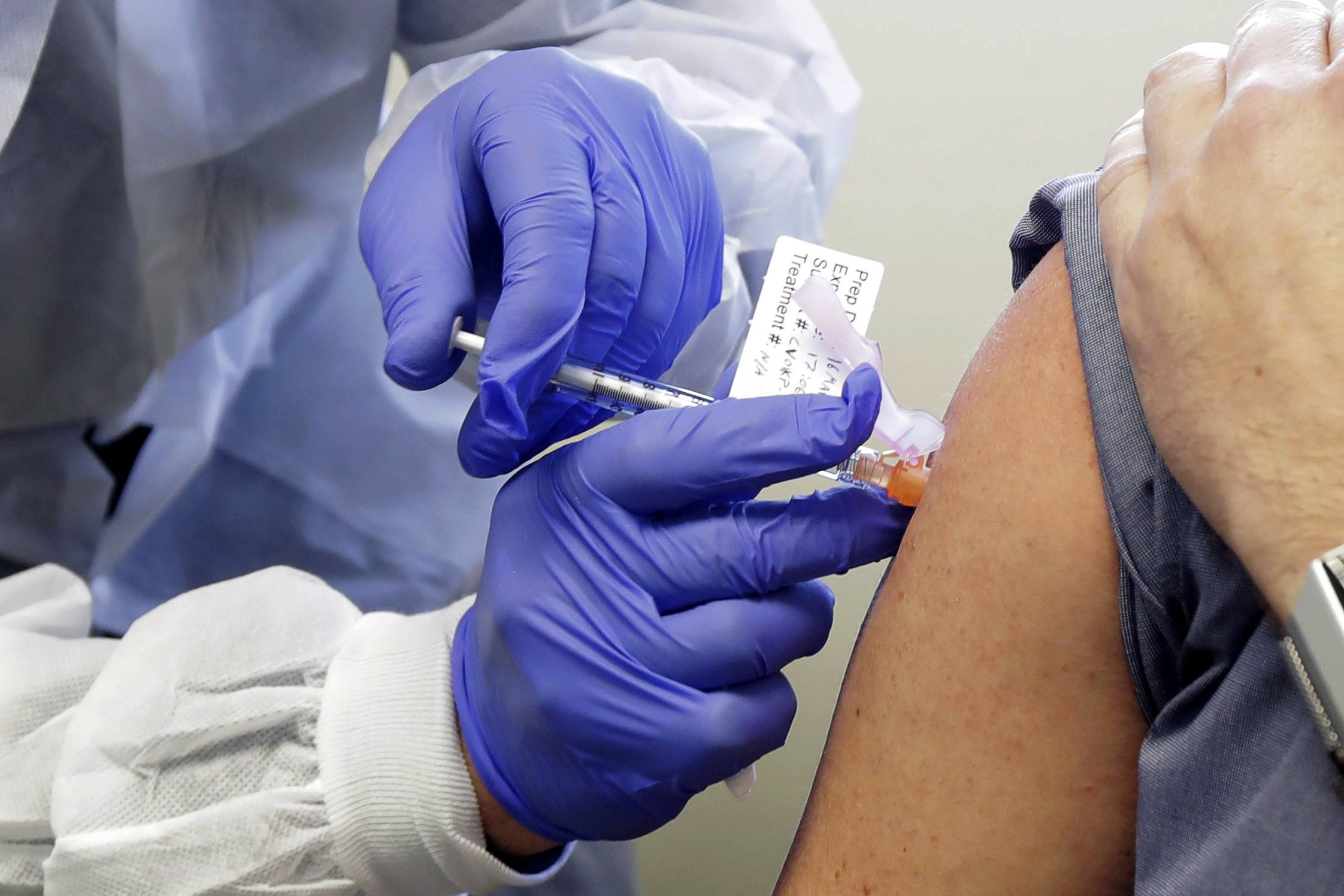
Regulatory authorities are gearing up for a deluge in people reporting side effects when the new COVID-19 vaccines go into use.
Even as vaccines like the one from Pfizer Inc. and BioNTech SE reach safety milestones and look set for regulatory approval, managing the reporting and follow-up of what are known as adverse drug reactions will be critical to keeping to the high levels of public participation needed for a vaccination program to be successful.
The U.S. Centers for Disease Control and Prevention plans to send daily texts to those who are vaccinated for the first week and then weekly texts for six weeks, while the Food and Drug Administration will also be monitoring side effects in real time.

















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.