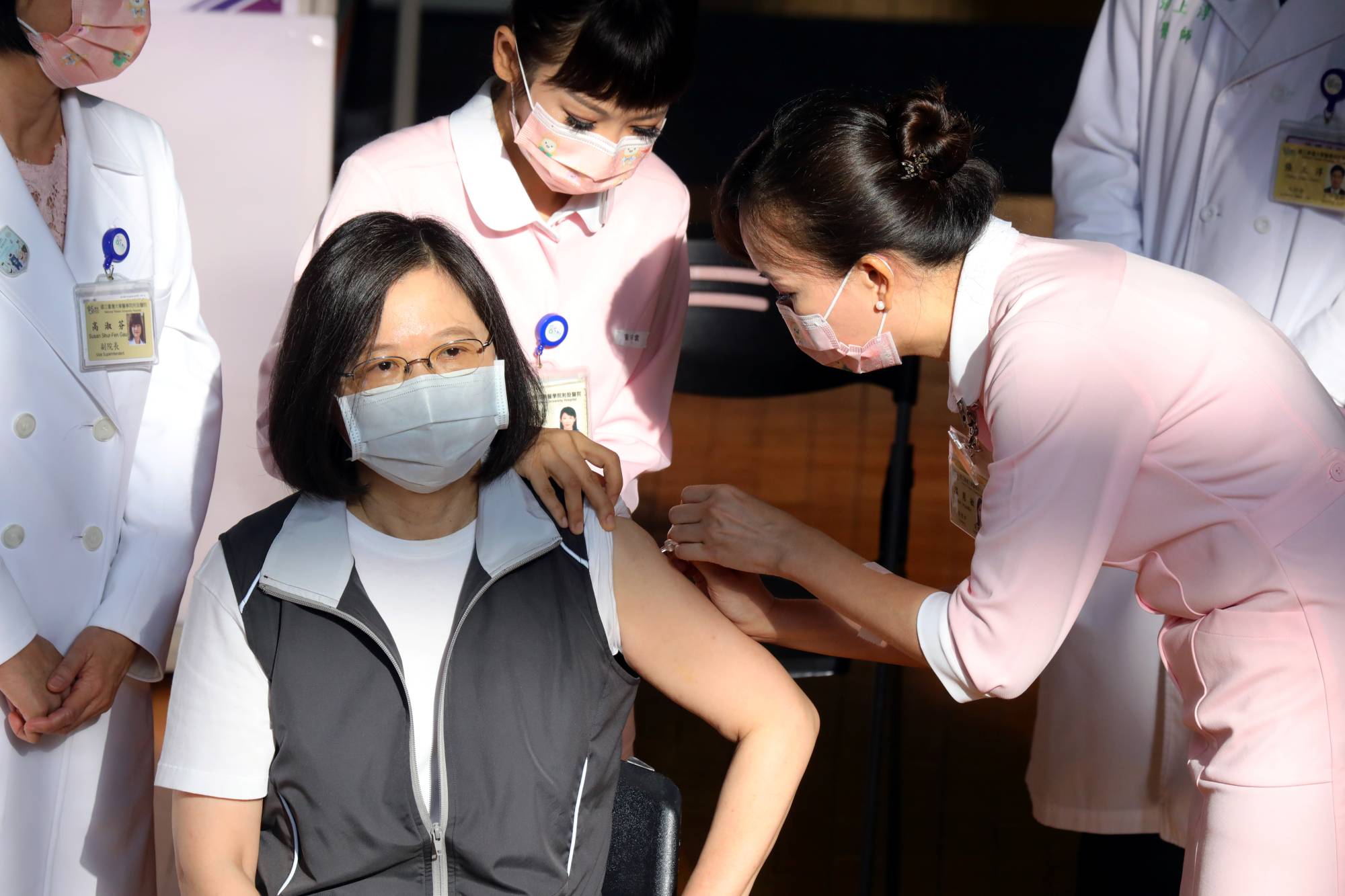
President Tsai Ing-wen got vaccinated with Taiwan's first domestically developed COVID-19 vaccine on Monday, giving her personal stamp of approval as the island begins rolling out the shot whose approval critics say has been rushed.
The health ministry last month approved the emergency use of Medigen Vaccine Biologics Corp.'s COVID-19 vaccine, part of a broader plan for inoculation self-sufficiency as delays in vaccine deliveries from global drug companies have affected Taiwan and many other countries.
Tsai, who had held off using vaccines from Moderna Inc. or AstraZeneca PLC, the current mainstay of Taiwan's vaccination program, received her Medigen shot at a hospital in central Taipei, demonstrating her confidence in the safety of the vaccine.


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.