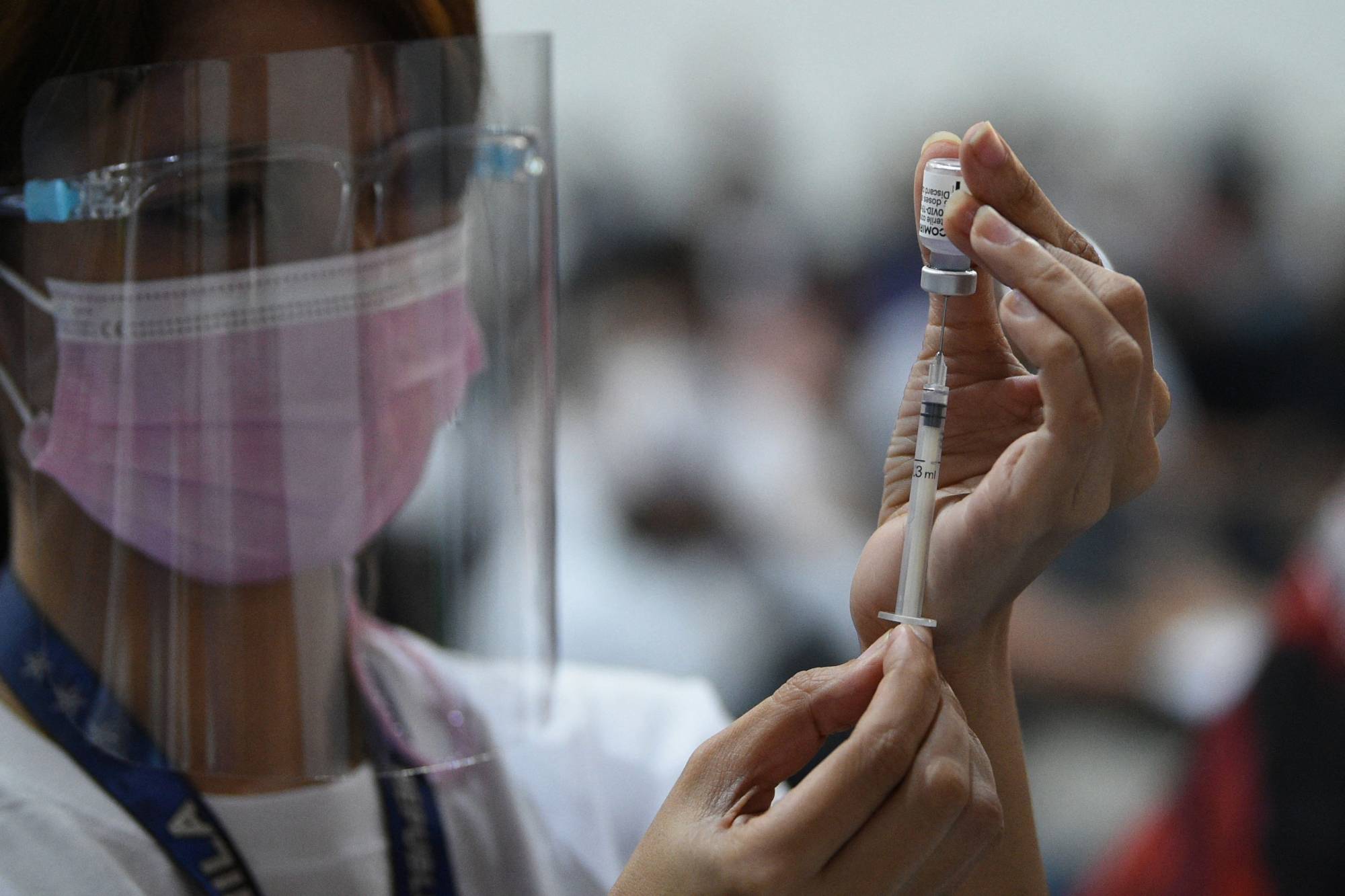
The latest surge in COVID-19 cases is widening one of the biggest inequities of the pandemic era: The gap between nations that have messenger RNA vaccines and those that don’t.
The cutting-edge technology, which made its debut during the pandemic, has proven more effective than any other in staving off infections and serious illness from the coronavirus. Yet just a handful of facilities in the U.S. and Western Europe account for almost all of the world’s mRNA vaccine supply — leaving many countries in a desperate race to catch up.
From South Korea to Indonesia and South Africa, governments are seeking to set up mRNA production and develop other collaborations in the space, but they’re also realizing it could take them years to master the new science, leaving their countries reliant on less potent vaccines that potentially expose them to repeated waves of COVID-19 and delay their economies from reopening.


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.