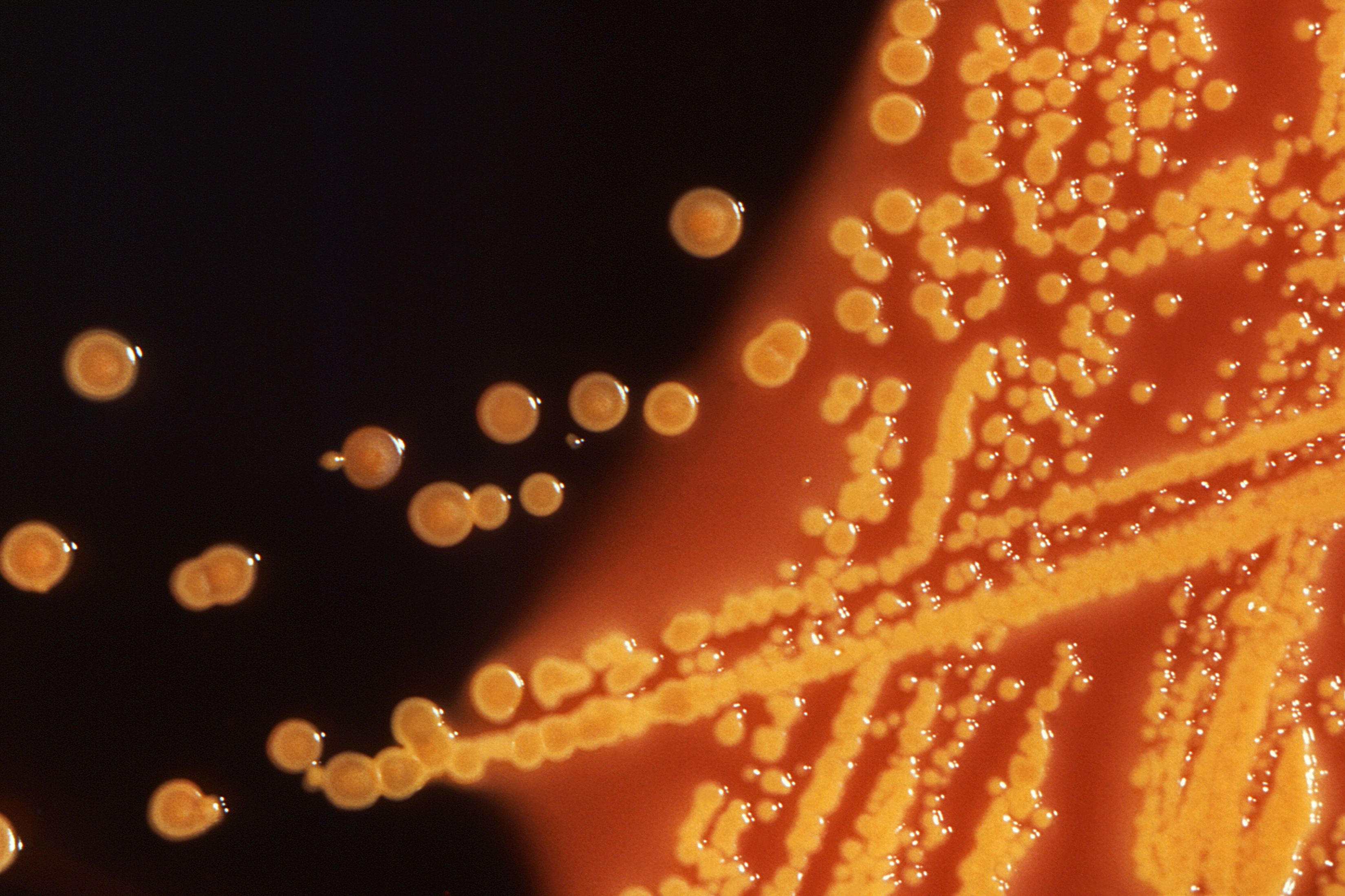
Drugmakers are renewing efforts to develop medicines to fight emerging antibiotic-resistant bacteria, but creating new classes of drugs on the scale needed is unlikely to happen without new financial incentives to make the effort worth the investment, companies and industry experts said.
American military researchers on Thursday announced the first U.S. case of a patient with an infection found to be resistant to the antibiotic colistin, a drug often held in reserve for when all else fails.
That put a spotlight on the urgent need for new medicines that can combat what health officials have called "nightmare bacteria."


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.