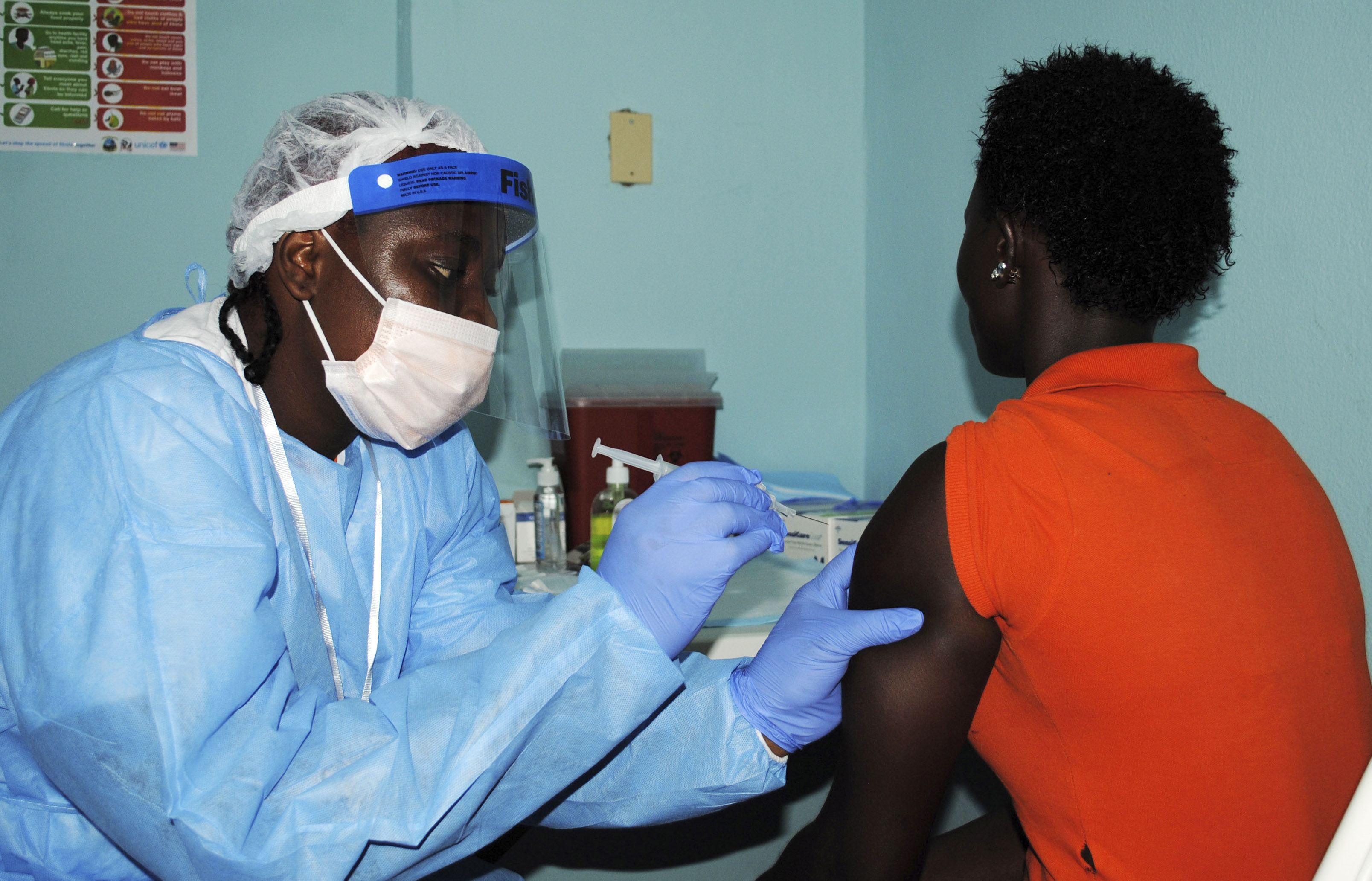
Liberia began a trial of experimental Ebola vaccines on Monday, involving thousands of volunteers as part of an effort to slow the spread of the deadly fever and prevent future outbreaks.
The epidemic has killed more than 8,900 people in West Africa since it began more than a year ago, overwhelming weak health care systems in Guinea, Liberia and Sierra Leone. Its spread now appears to be slowing, especially in Liberia, which has reported just 15 new cases in the past 21 days.
The trial to test two vaccines from GlaxoSmithKline and New Link/Merck began at the government-run Redemption Hospital in Monrovia, a cluster of cement blocks in the teeming New Kru Town neighborhood that was one of the first parts of the capital to be struck by the disease.


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.