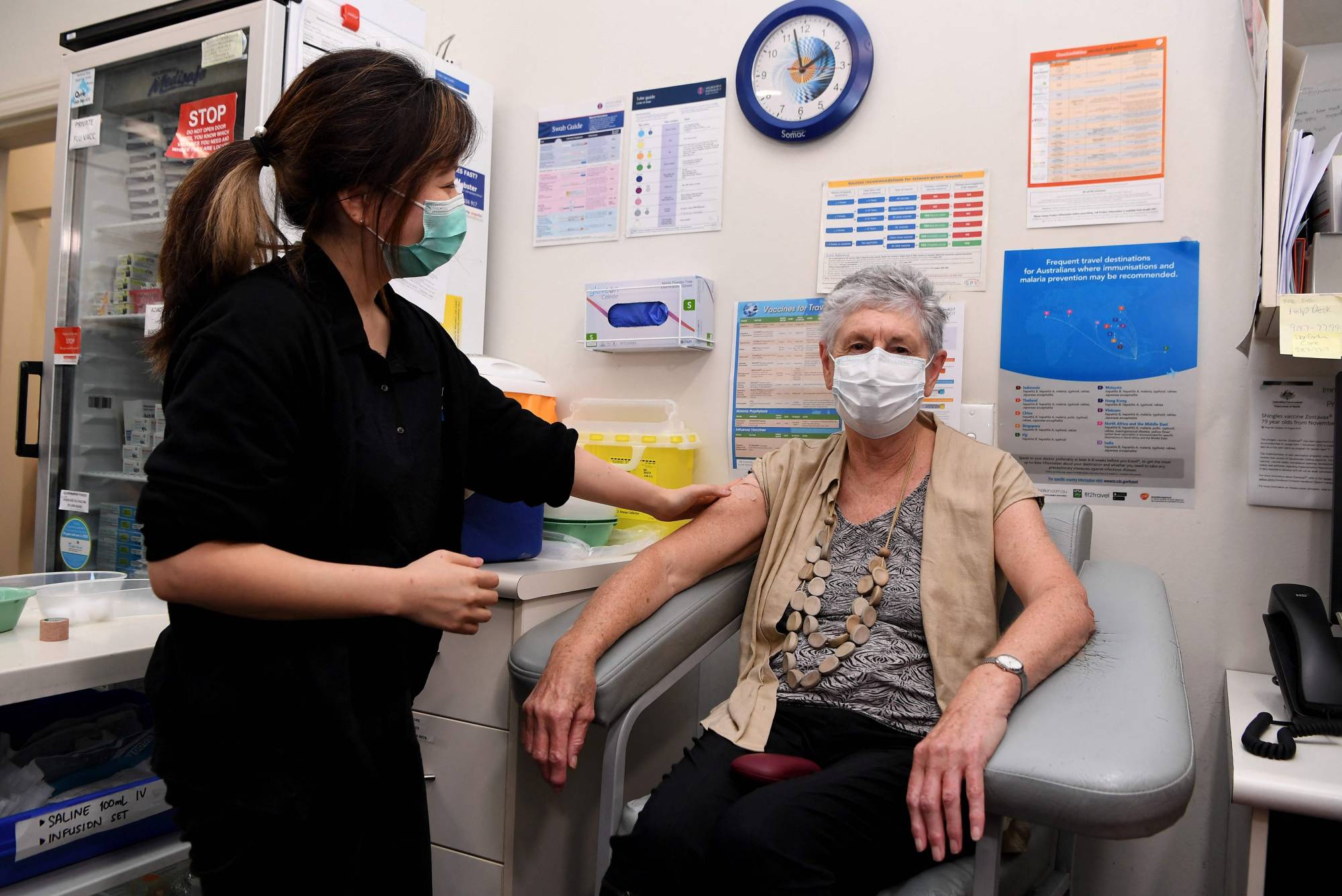
Countries across the Asia Pacific are demurring on AstraZeneca PLC’s COVID-19 vaccine, or restricting its use, in moves that may delay protection for the region amid concerns that the shot could trigger a rare and potentially deadly blood clotting condition.
After guiding against the vaccine’s use in people under 50 and saying that it didn’t expect shipments from Europe to arrive on time, Australia’s government abandoned a pledge that everyone in the country would receive a first dose by October.
In Hong Kong, officials said they wouldn’t take delivery of the vaccine supply they’ve ordered from the British drugmaker this year, as the city plans to rely on vaccines from BioNTech SE and Sinovac Biotech Ltd.


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.