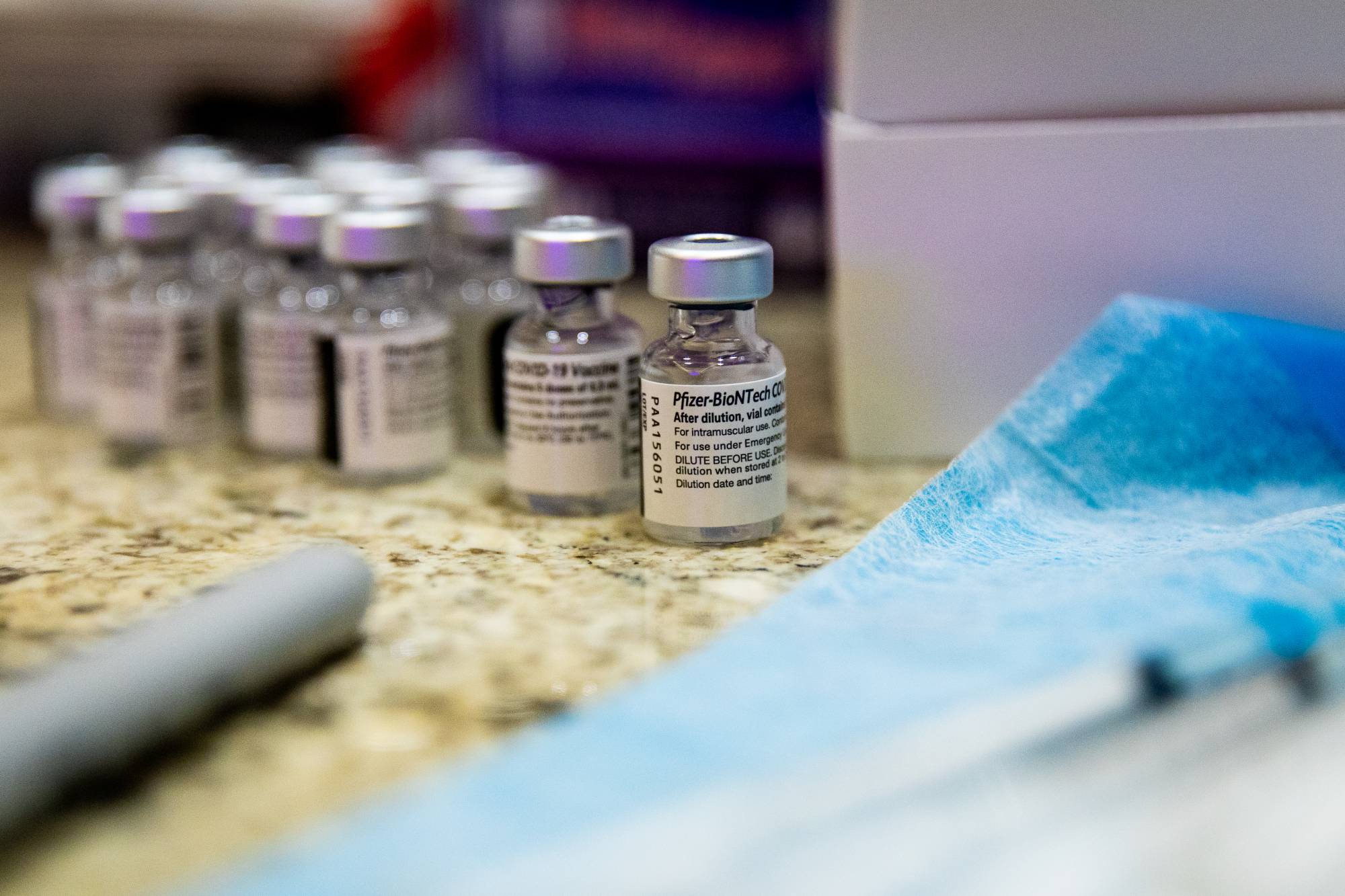
U.S. drugmaker Pfizer Inc.’s mRNA vaccine against COVID-19 has become the first to be formally approved for use in Japan, paving the way for inoculations to start from Wednesday, but significant challenges still lie ahead for local governments that must set up the necessary logistics for a smooth vaccine rollout.
Pfizer’s novel coronavirus vaccine got the final OK for emergency use from the health ministry on Sunday, two days after a government committee gave the shot its seal of approval.
Prime Minister Yoshihide Suga said the vaccine rollout would begin Wednesday with front-line health care workers.


















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.