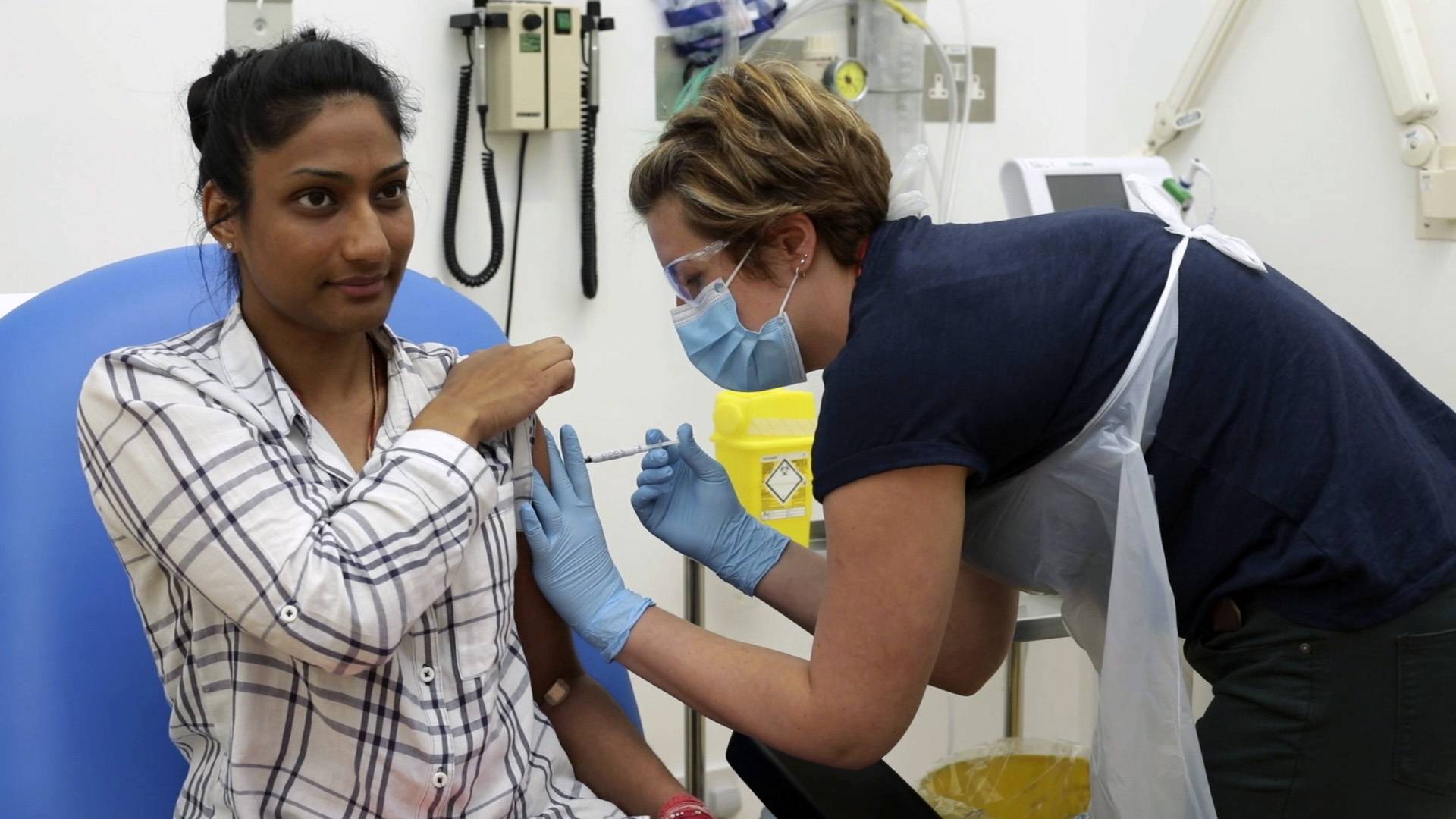
The notion of testing COVID-19 vaccines by deliberately infecting volunteers with the novel coronavirus — something that’s now on the table — isn’t necessarily crazy or unethical. It’s smart, and has benefits that far outweigh the risks.
Challenge trials, as scientists call them, vastly speed up the process of vaccine testing and might also help us understand natural immunity of people who have already been infected.
This doesn’t mean abandoning the framework of medical ethics that protects human research subjects from abuse. It means recognizing that there are risks and benefits unique to this situation.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.