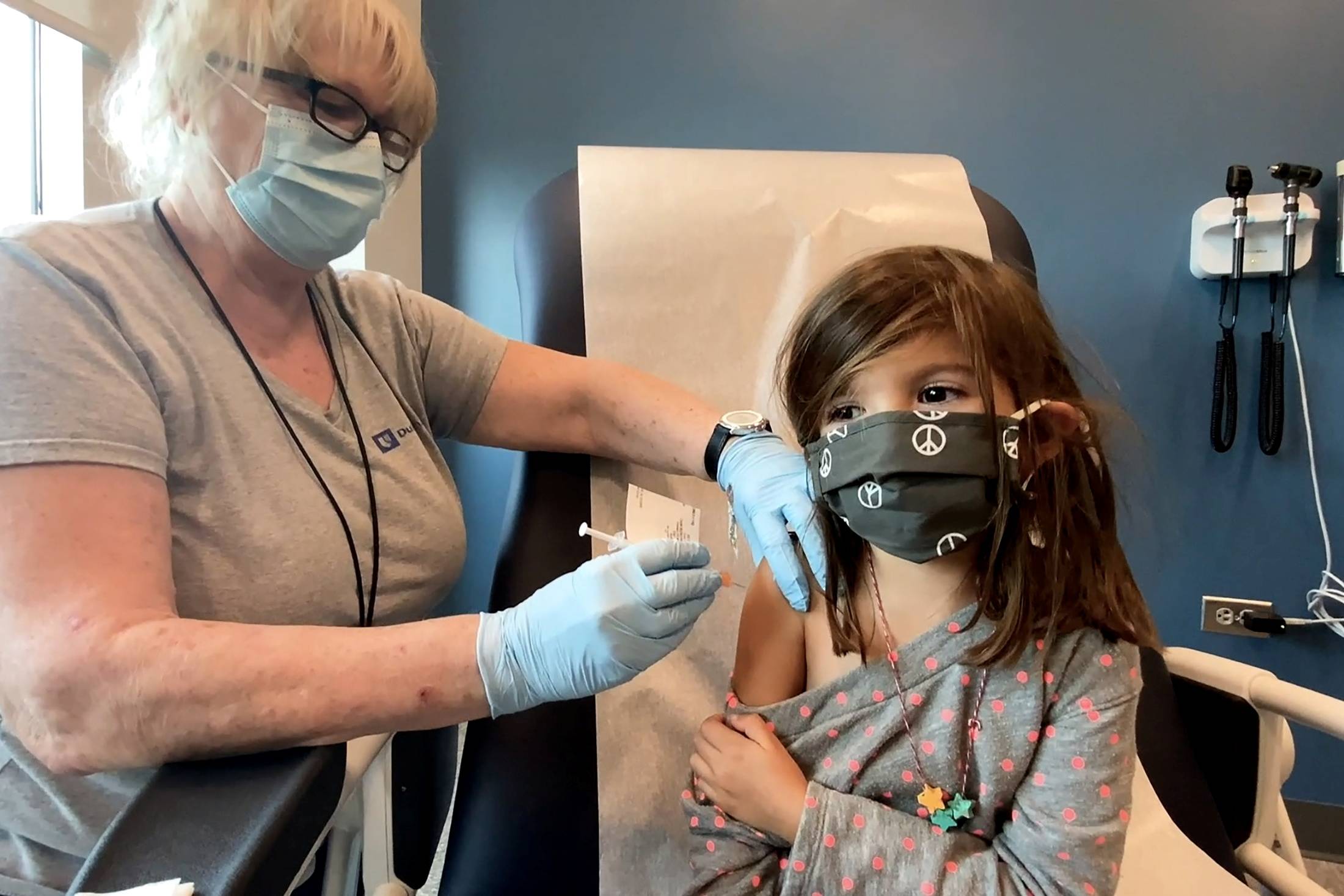
U.S. drugmaker Pfizer Inc.'s Japan unit said Thursday it is in talks with the government about applying for approval of its COVID-19 vaccine for use in children age 5 to 11.
The move follows the overwhelming backing on Tuesday by the U.S. Food and Drug Administration’s advisory committee for lowering the minimum age for the vaccine from the current 12 to 5, paving the way for formal authorization for the shot's use in 28 million children there from early November.
Pfizer said the company is in talks with regulatory authorities in Japan but added it cannot comment on the timing or other details of the application or its approval.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.