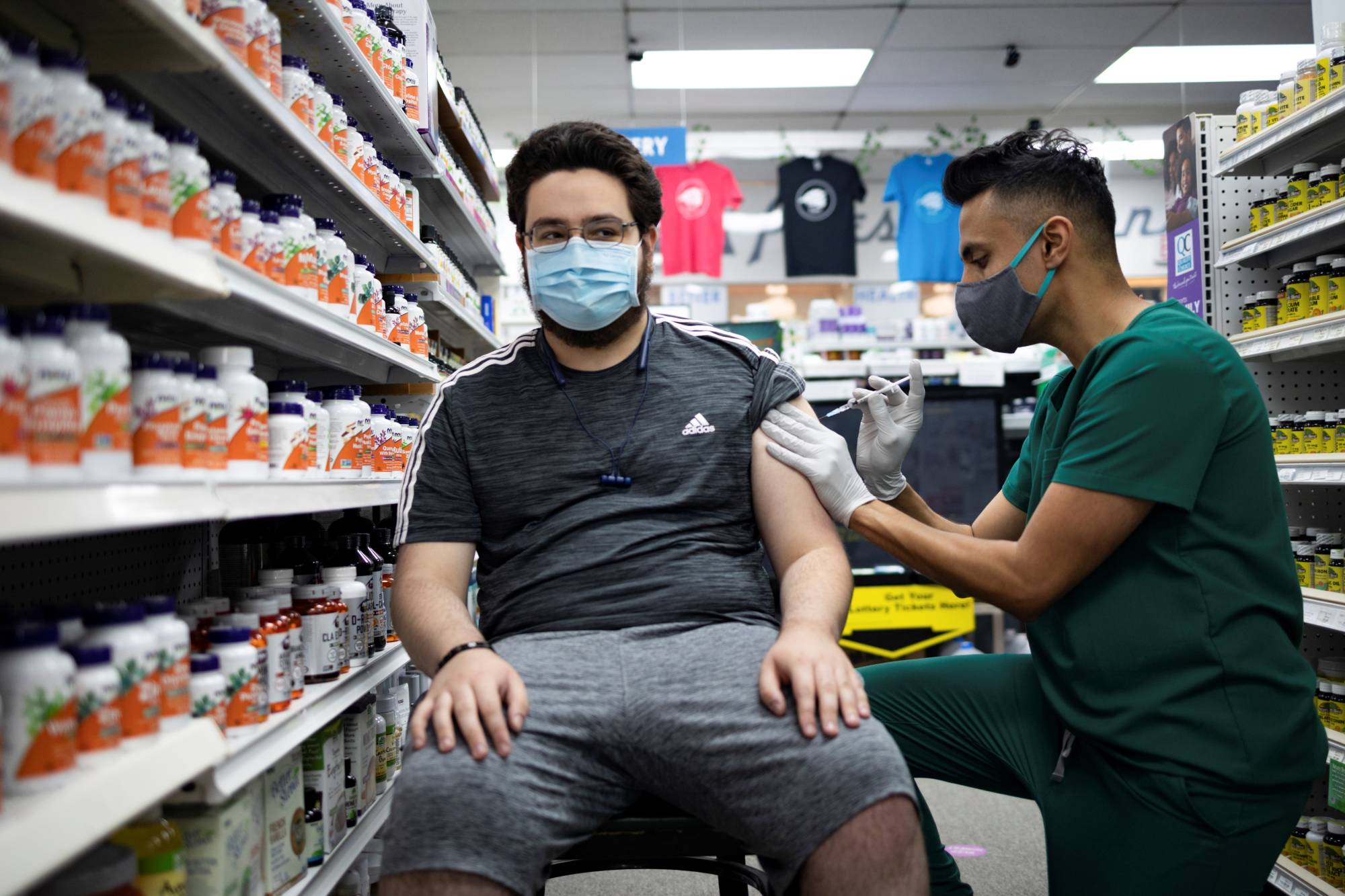
The Biden administration has decided that most Americans should get a coronavirus booster vaccination eight months after they received their second shot, and could begin offering third shots as early as mid— to late September, according to administration officials familiar with the discussions.
Officials are planning to announce the decision as early as this week. Their goal is to let Americans who received the Pfizer-BioNTech or Moderna vaccines know now that they will need additional protection against the delta variant that is causing caseloads to surge across much of the nation. The new policy will depend on the Food and Drug Administration’s authorization of additional shots.
Officials said they expect that recipients of the Johnson & Johnson vaccine, which was authorized as a one-dose regimen, will also require an additional dose. But they are waiting for the results of that firm’s two-dose clinical trial, expected later this month.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.