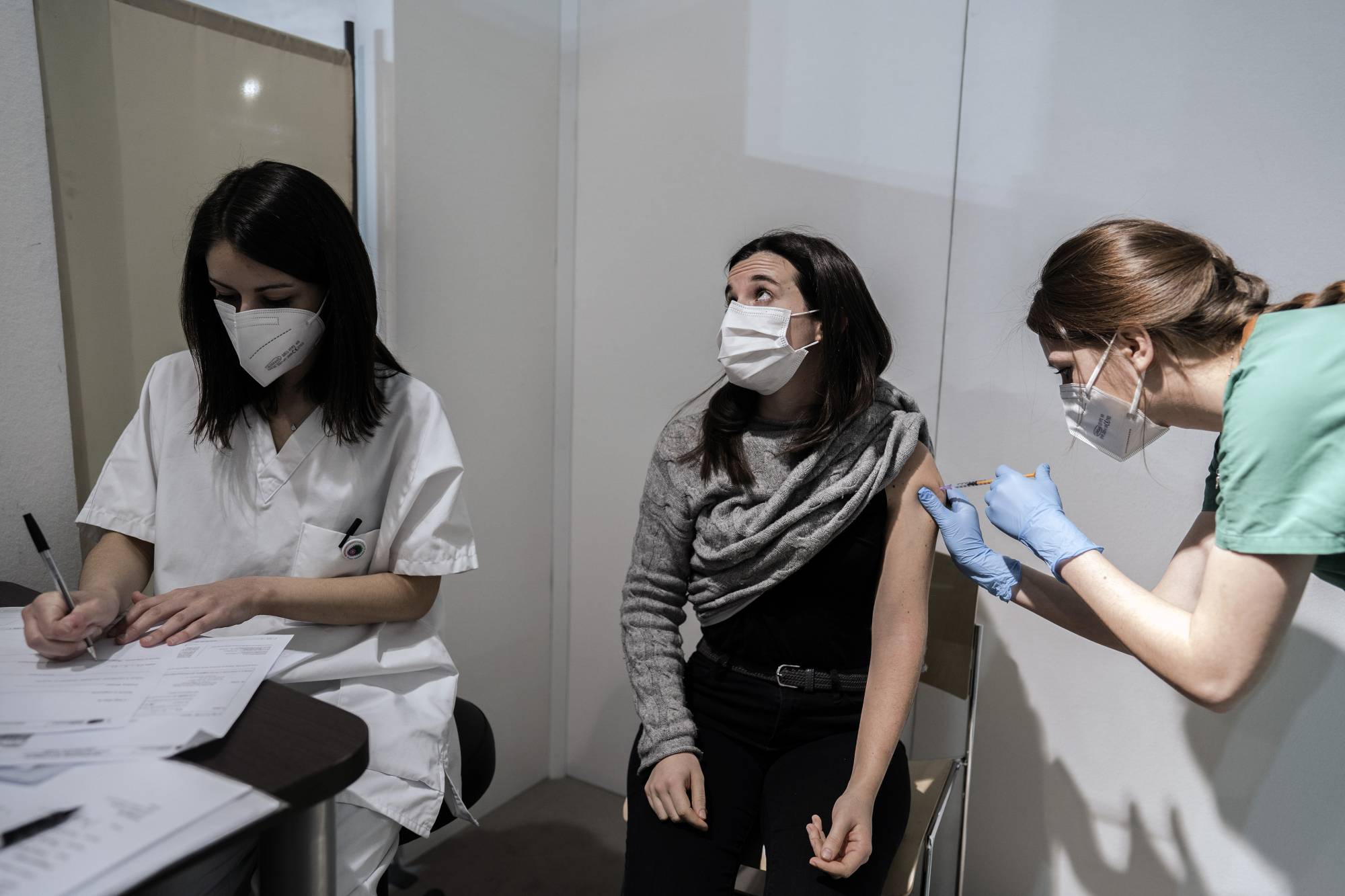
AstraZeneca PLC may have released outdated information about its COVID-19 vaccine trial, giving an "incomplete” view of the efficacy of the shot, said the leading U.S. agency on infectious disease.
The Data and Safety Monitoring Board, charged with ensuring the safety and accuracy of AstraZeneca’s vaccine trial, has expressed concerns to the National Institute for Allergy and Infectious Diseases that the information released about the testing results included outdated information.
This "may have provided an incomplete view of the efficacy data,” the agency said in a statement early Tuesday, without elaborating.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.