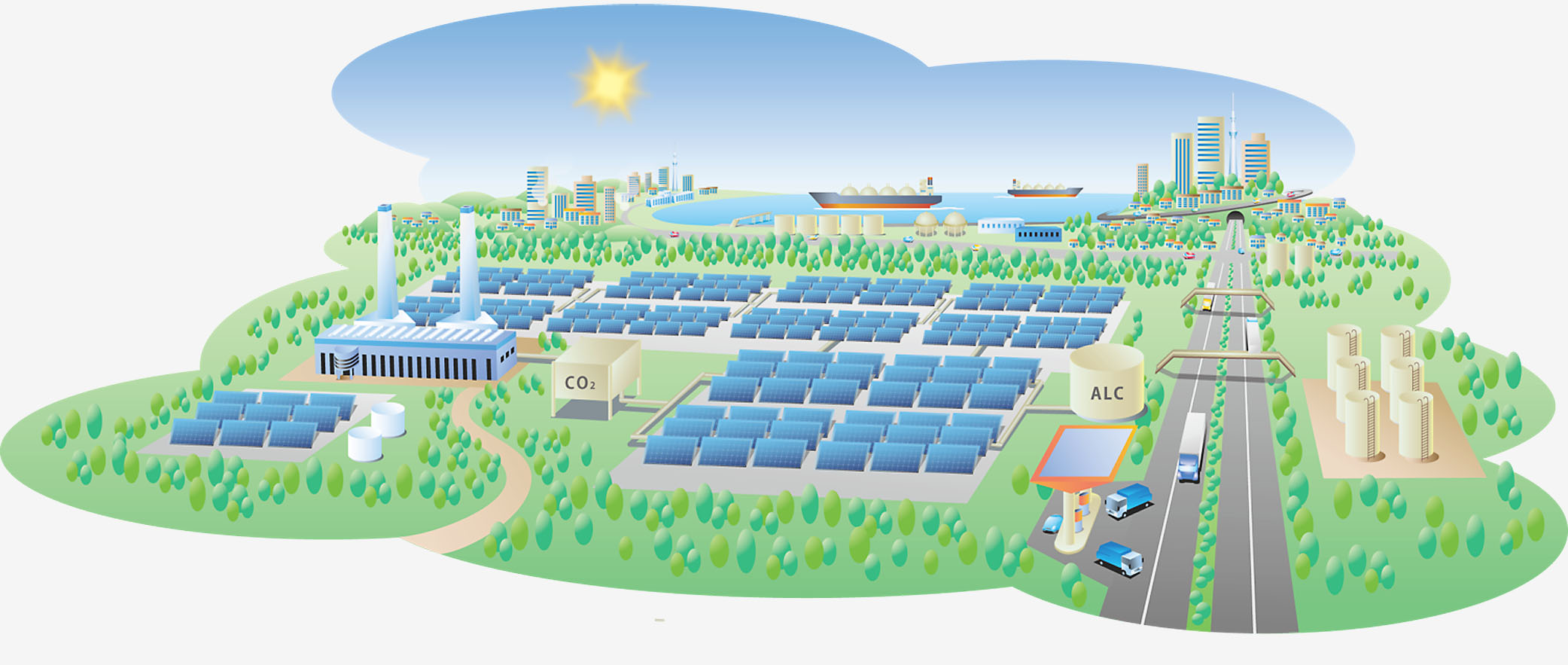
The world's fight against global warming would be dramatically easier if a cheap technology was available to turn carbon dioxide into something useful.
In fact, researchers and firms are trying to do just that with artificial photosynthesis, a process that uses sunlight, water and carbon dioxide to produce oxygen and substances that can be turned into energy, just as plants and algae do via chlorophyll.
Expectations in this field are rising because the technology might help meet globally rising demand for clean energy and, more importantly, reduce carbon dioxide emissions.
















With your current subscription plan you can comment on stories. However, before writing your first comment, please create a display name in the Profile section of your subscriber account page.