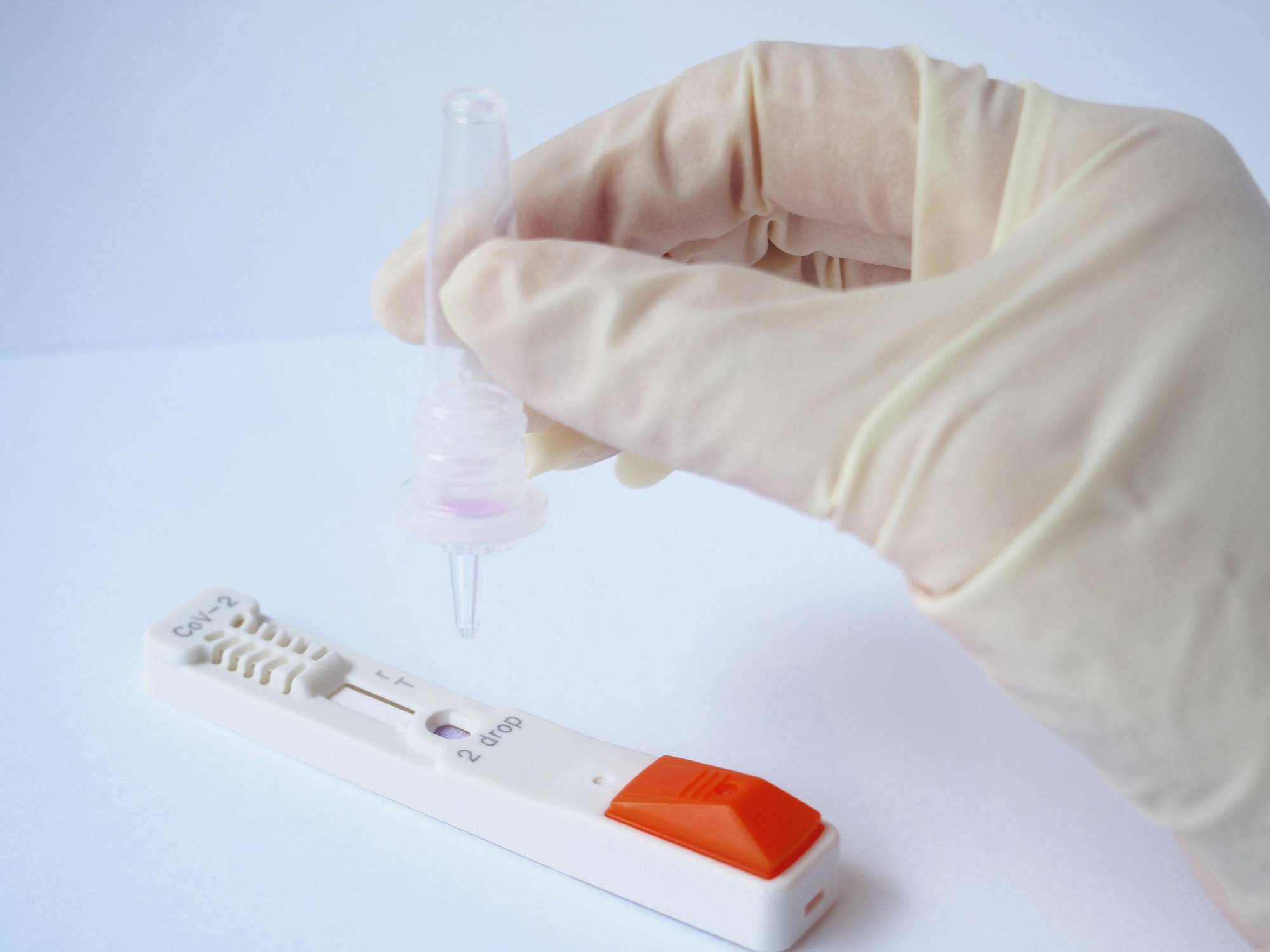
The Japanese government approved on Wednesday test kits that can detect novel coronavirus antigens in 15 to 30 minutes, in the hope of improving its testing regime amid growing demand for a simpler and faster method.
The step comes as the currently dominant polymerase chain reaction, or PCR, test requires a few hours at least to produce a result, and there has been a rise in the number of people who need testing in the country.
Fujirebio Inc., the test kit producer, has said it can supply 200,000 kits a week and will consider expanding output if there is more demand.
In antigen tests, commonly used for testing for the flu, doctors insert a swab into a patient's nostril and get the results on site.
The test detects proteins unique to the virus and does not need to be conducted at labs like the PCR test, which involves amplifying small amounts of DNA sequences of the virus. In some cases, patients have waited a week before receiving their PCR test result.
But as the antigen test is considered to have lower sensitivity to the virus than the PCR test, people with negative results will also be required to undergo PCR tests to ensure accuracy.
The quick testing method is expected to be used to test patients who need immediate treatment, and to check for infection within medical institutions and nursing homes.
The test is expected to be covered by Japan's public insurance system and patients will not be required to pay anything out-of-pocket if they undergo the test on the advice of a doctor.
The government also started distributing the anti-viral drug remdesivir, developed by U.S. biopharmaceutical company Gilead Sciences Inc., to hospitals this week to treat patients with severe symptoms of COVID-19.